In a late-night announcement on Monday, Prime Minister Scott Morrison declared all Australians, no matter their age, will be able to get their AstraZeneca jab at GP clinics under an indemnity scheme.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
However, the health advice on the vaccines from Australian Technical Advisory Group on Immunisation (ATAGI) and the Australian Medical Association (AMA) has not changed. These entities state the preferred vaccine for people under 60 is Pfizer.
Federal Minister for Health Greg Hunt said on Tuesday Australia had passed 7.5 million vaccinations including 5.95 million first-dose vaccinations and over 126,000 vaccinations.
"AstraZeneca remains the preferred vaccine for people 60 years and over; that has not changed," he said.
"The advice of ATAGI is that Pfizer is the preferred for people under 60 and the clinical advice of ATAGI.
"However, as has always been the case - and I think it is very important to emphasise this [is] on the basis of informed consent."
Read more:
- New COVID-19 roadmap - here's how Scott Morrison says we 'return to normal'
- Victoria records zero COVID-19 cases overnight, 30 active cases
- What you need to know about the COVID-19 Delta variant
- Bendigo doctor says AstraZeneca supply still available
- Bendigo doctors roll-up sleeves for second AstraZeneca vaccination
- More COVID-19 vaccinations will lower lockdown risk: Bendigo doctor
The Victorian state-run vaccine operation has provided indemnity for anyone already eligible to be vaccinated; whether they're over 40 years, an eligible frontline worker or otherwise immunocompromised, with the AstraZeneca vaccine.
"We want to see every Victorian vaccinated as soon as possible.," a Victorian government spokesperson said.
"We're continuing to work with the Commonwealth to clarify its position on vaccine eligibility and what this decision means for state-run vaccine clinics.
"We urge people to have a discussion with their GP to make an informed decision on their vaccine choice based on their individual health circumstances."
The Bendigo Advertiser spoke to Bendigo Primary Care Centre clinical director Sheriden Emonson and Bendigo Health to answer some of your questions.
What exactly has changed?
While Mr Hunt said Pfizer was preferred for people under 60, he said individual patients could make the decision to take up AstraZeneca - in consultation with their doctor - based on their circumstances and judgement.
"The difference is that the access for those who are of all ages, for the AstraZeneca vaccine, will be available through general practices," Mr Hunt said.
"[There is an] update to the GPs which is going out during the course of today and also the change to the Medicare item which will now allow over the coming days for everybody under 50 also to seek a vaccine consultation if they so desire."
ATAGI says Pfizer "is preferred" over AstraZeneca for people aged 16 to 60 years.
"The benefits (of having AstraZeneca if you are under 60) are likely to outweigh the risks for that individual (if) the person has made an informed decision based on an understanding of the risks and benefits."
I'm under 40, should I book in for an AstraZeneca shot?
AstraZeneca remains recommended for people aged over 60.
Mr Hunt confirmed on Tuesday that medical advice had not changed, leaving Pfizer as the preferred vaccine for under 60s.
It was despite Mr Morrison's statement on Monday that the indemnity scheme would allow Australians under 40 to receive AstraZeneca.
Victorian Health Minister Martin Foley said the AstaZeneca decision was not made at national cabinet. Mr Foley said he had written to the federal government seeking further advice.
Should I wait for Pfizer?
Dr Emonson said the current vaccine options available to Australians were between Pfizer and AstraZeneca and people should weigh up their options.
"We need to weigh up the potential risks of a vaccine versus potential risk of COVID-19 outbreak in our community. We want to be protected against the coronavirus," she said.
"The Department of Health states that if you received your first dose of AstraZeneca COVID-19 vaccine and did not experience abnormal side effects, ATAGI recommends you still receive your second AstraZeneca dose.
"The best way to get to a new normal is to be immune and at the moment we have Pfizer and AstraZeneca."
We have removed our paywall from this story about the coronavirus. If you would like to support our journalists, subscribe here.
What is the chance of getting TTS (blood clots) from AstraZeneca?
ATAGI stated from early April to 16 June 2021, 60 cases of confirmed or probable thrombosis and thrombocytopenia syndrome (TTS) have been reported in Australia.
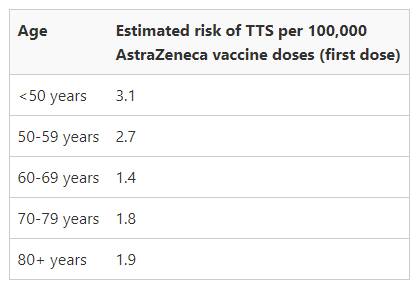
ATAGI said the risk of TTS following a second dose of COVID-19 Vaccine AstraZeneca is much lower than the risk following a first dose.
Meanwhile, the estimated risk of getting a blood clot per year from being on the contraceptive pill is one in 2,500, or 40 in 100,000 (almost 13 times more likely than getting a blood clot from AstraZeneca for people aged under 50).
What side effects might I get?
The Australian Government Department of Health states people may have some temporary side effects after receiving a COVID-19 vaccine, the same as receiving any vaccine (like the flu shot).
Common side effects after AstraZeneca include:
- injection site pain or tenderness
- tiredness
- headache
- muscle pain
- fever and chills
Most side effects are mild and temporary, going away within one or two days.
As with any medicine or vaccine, there may be rare and/or unknown side effects.
Dr Emonson, 44, received her second dose of AstraZeneca on Saturday. She said she had no major adverse reaction to either of her shots.
What could the chat with my GP look like?
Dr Emonson encouraged people to look at all the facts and make the right choice for themselves and the community about getting vaccinated.
"As GPs our position is really important to speak to people and help them feel empowered to make the right decision for them," she said.
"We encourage people to think about themselves, their loved ones and getting back to a new normal.
"We want people to weigh these thoughts and then make the decision about the vaccines."
Who is an essential worker for the jab?
The Australian Government Department of Health states that essential workers for the 16 to 39 age group are:
- Quarantine and border workers
- Aged and disability care workers
- Disability care residents
- Frontline healthcare workers
- Other healthcare workers
- Household contact of quarantine and border workers
- Critical and high risk workers
- Traveller with a travel exemptions
- Aboriginal or Torres Strait Islander peoples
- Person with an underlying medical conditions
- Other eligible individuals
Should I mix vaccinations?
The Australian Government Department of Health states for greater coverage against coronavirus, if you've had your first dose of the AstraZeneca vaccine, and did not experience abnormal side effects, ATAGI recommends you still receive your second AstraZeneca dose.
What do I do if I'm pregnant, breastfeeding or trying for a baby?
ATAGI has recommended the Pfizer vaccine at any stage of pregnancy or while breastfeeding.
A Bendigo Health spokesperson confirmed the findings, which were released at the beginning of June.
"ATAGI has ruled the Pfizer vaccine is safe for pregnant women," the spokesperson said.
"You are also safe to get vaccinated if considering getting pregnant or breastfeeding."
The Royal Australian and New Zealand College of Obstetricians and Gynaecologists (RANZCOG) said pregnant women have been advised that the Pfizer vaccine is safe to have with "no evidence suggesting an increased risk of miscarriage or teratogenicity".
The Bendigo Advertiser sent the following questions to the Australian Department of Health to answer:
Can you explain how the COVID-19 Vaccine Claims Scheme (indemnity waiver) works?
The COVID-19 Vaccine Claims Scheme will be backdated to the start of the national vaccine rollout - February 22, 2021 - and will be linked to the Human Biosecurity Emergency Period under the Biosecurity Act 2015. It is planned to be a time limited, targeted measure to support increased vaccination uptake and protect Australians.
It will provide coverage for patients and health professionals administering or receiving a Therapeutics Goods Administration (TGA) approved COVID-19 vaccine.
The establishment of a COVID-19 Vaccine Claims Scheme (the scheme), will support claims being made against privately practising health practitioners who administer a COVID-19 vaccine approved for use by the Therapeutic Goods Administration (TGA).
This new fit-for-purpose medical indemnity scheme will provide a patient centric approach through a portal which will guide potential claimants on the most appropriate pathway under the scheme. It will provide an additional level of assurance to patients by ensuring doctors and other practitioners administering or advising on COVID vaccines as part of the Commonwealth vaccination program have appropriate direct indemnity coverage. It will also aim to simplify patient access to compensation related to vaccine manufacturers' indemnity.
It is proposed that a single portal will operate to give support to people who think they have suffered a significant adverse health outcome or economic loss as a result of being vaccinated. Substantiated claims will be able to receive no fault compensation without undue red tape or the need for a formal court process.
Final scheme design is to be resolved and will include consultation with key stakeholders in the coming weeks.
To further support the choice of people considering their vaccine options, the Government is extending two Medicare items that allow GPs and other medical practitioners to provide in-depth clinical assessments of a patient's individual risks and benefits. Already available for patients aged 50 years and over, the Medicare items 10660 and 10661 will be available for all patients, regardless of age, with effect from June 29.
All Australians are being supported and encouraged to make an informed decision before consenting to COVID-19 vaccination and it is an individual's decision as to whether they wish to discuss COVID-19 vaccination with a GP. The Government has implemented a number of MBS items, which must be bulk-billed in-line with the Government's commitment that all Australians will be able to be vaccinated for free, to support discussions about risks and benefits of vaccination and informed consent. General practices have implemented different models of managing these consultations and vaccination and it is recommended that patients discuss appropriate appointment arrangements with their practice.
I'm under 40 and I've registered to receive the vaccine. Will I still need to see a GP even when the roll-out (eg: 1B) says I'm eligible for the vaccine?
No, you do not need to see your GP if you are eligible for Pfizer in a priority group.
Is there still a priority list for essential workers to get the vaccine before people who are under 40 and have been to a GP?
At National Cabinet on June 21, all states and territories agreed to prioritise Pfizer appointments for people aged 40-59 and people in priority groups who are aged under 40 years.
Why is the federal government opening AstraZeneca when it was reported last week it would be phased out?
In a press conference on June 23, Lieutenant General John Frewen said he expected there would be AstraZeneca supply until the end of the year if needed.
"The assumptions that are in the ranges there are that for all of those people and cohorts that AstraZeneca is preferred," he said. "We think they (the people who are in the allocated groups to receive AstraZeneca) will have received their AstraZeneca before the fourth quarter.
"For any people who still do require AstraZeneca, we will have allocations available into the fourth quarter and we fully expect that there'll be adequate supply of AstraZeneca well into the fourth quarter."
Are there other types of COVID-19 vaccine that will be delivered soon in Australia?
On June 24 the Therapeutic Goods Administration, part of the Department of Health, granted provisional determination to Moderna Australia Pty Ltd in relation to the COVID-19 Vaccine, Elasomeran.
The Australian Government has secured 25 million doses of the Moderna COVID-19 vaccine. The agreement includes the supply of 10 million doses in 2021 and of 15 million doses of Moderna's updated variant booster vaccine in 2022.
Where can I get vaccinated?
Bendigo Health's Mass Vaccination Clinic is located at 113-133 Mollison Street, Bendigo and open from 10am to 5pm, seven days a week.
Bendigo Health is currently administering the Pfizer vaccine for those in priority 1a and 1b groups aged 16 to under 60 years and the AstraZeneca vaccine to those over 60 years.
For people aged 50-59 and have had their first dose of AstraZeneca, they are encouraged to get your second dose.
Walk-in appointments at the Mollison Street centre for Pfizer have been paused until further notice but people with booked appointments will be vaccinated.
Register for the vaccine via bendigohealth.org.au/registerforvaccine
Central Victorian GPs have also been supplied with vaccines. Contact your GP to find out more.
With AAP.
For urgent questions, email Bendigo Health's coronavirus team via email COVIDVaccine@bendigohealth.org.au
Our journalists work hard to provide local, up-to-date news to the community. This is how you can access our trusted content:
- Bookmark bendigoadvertiser.com.au
- Make sure you are signed up for our breaking and regular headlines newsletters
- Follow us on Twitter @BgoAddy
- Follow us on Instagram @bendigoadvertiser
- Join us on Facebook
- If you're a subscriber, join our subscriber-only Facebook group.
- Follow us on Google News


